USFDA Approves Duvyzat for Muscular Dyst
USFDA has approved Italfarmaco’s Duvyzat (givinostat) oral medication for treatment of Duchenne Muscular Dystrophy (DMD).

Warning letters, 483s, Recalls, Import Alerts, Audit observations

USFDA has approved Italfarmaco’s Duvyzat (givinostat) oral medication for treatment of Duchenne Muscular Dystrophy (DMD).

USFDA has approved Idorsia’s new drug Tryvio, a New Molecular Entity (NME) drug Aprocitentan for
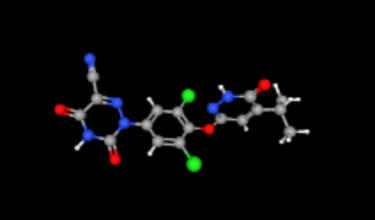
FDA has approved Madrigal Pharmaceuticals’ Rezdiffra (Resmetirom) for treatment of fatty liver disease (Noncirrhotic Nonalcoholic
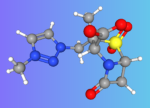
Indian Pharma company Orchid Pharma’s New Molecular Entity (NME) Enmetazobactam gets USFDA approval for treatment

Vamorolone is a synthetic corticosteroid approved by USFDA for the treatment of Duchenne Muscular Dystrophy