Field Alert Reports (FAR-USFDA); Europe
USFDA Field Alert Reports; USFDA Field Alert Report Submission: Questions and Answers Guidance for Industry;

Warning letters, 483s, Recalls, Import Alerts, Audit observations
USFDA Field Alert Reports; USFDA Field Alert Report Submission: Questions and Answers Guidance for Industry;
Excipient Risk Assessment (Europe): Guidelines of 19 March 2015 on the formalised risk assessment for

Qvents compilation of regulatory guidelines on Data Integrity : FDA guidance for design, operation, and
USFDA Controlled Correspondence Related to Generic Drug Development USFDA Guidance Document – Controlled Correspondence Related

USFDA: Investigating Out-of-Specification (OOS) Test Results for Pharmaceutical Production – Level 2 revision MHRA: Out-of-specification
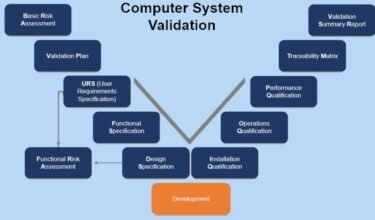
Qvents compilation of regulatory guidance’s on Computer System Validation, Electronic Data Review from USFDA, EMA,