

Discussion forum for Pharma Quality events, Regulatory Actions
Warning letters, 483s, Recalls, Import Alerts, Audit observations

Warning letters, 483s, Recalls, Import Alerts, Audit observations
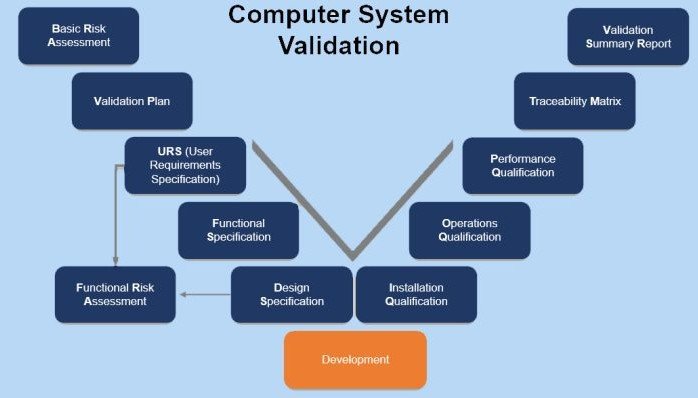
Qvents compilation of regulatory guidance’s on Computer System Validation, Electronic Data Review from USFDA, EMA, PIC-S. Access all guidelines on the topic – from USFDA 21 CFR Part 11 requirements, Questions and Answers, Document heavy CSV approach, Risk based frame work of CSA. Revised EC Annex 11 with focus on cybersecurity controls and EC Annex 22 focus on AI systems
Access all guidelines on the topic – from USFDA 21 CFR Part 11 requirements, Questions and Answers, Document heavy CSV approach, Risk based frame work of CSA. Revised EC Annex 11 with focus on cybersecurity controls and EC Annex 22 focus on AI systems