FDA Warning Letter to Simtra Biopharma F
The U.S. FDA has issued a Warning Letter to Simtra BioPharma Solutions following cGMP deviations

Warning letters, 483s, Recalls, Import Alerts, Audit observations

The U.S. FDA has issued a Warning Letter to Simtra BioPharma Solutions following cGMP deviations

The USFDA has issued a Warning Letter to the Indian drug manufacturing facility of Patcos
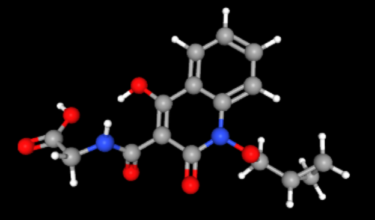
Indian pharma major Zydus Lifesciences has received approval from China’s National Medical Products Administration (NMPA)
The USFDA has Approved the NDA for ADQUEY (Difamilast 1%) ointment for the treatment of
The Government of India has published a draft notification proposing an amendment to the Drugs

The USFDA issued a Warning Letter to API manufacturer Chemspec Chemicals (Mumbai) in December 2025,

The USFDA has classified Sun Pharma’s Baska (Halol) facility (FEI: 3002957541) as Official Action Indicated
An USFDA 483 to CDMO major Catalent’s Baltimore facility (FEI 3015558590) in US cite poor

The USFDA has escalated the OAI (Official Action Indicated) classification of the Novo Nordisk–owned Catalent

FDA has published the USFDA Form 483 issued to sterile drug manufacturing facility of Fareva