

Discussion forum for Pharma Quality events, Regulatory Actions
Warning letters, 483s, Recalls, Import Alerts, Audit observations

Warning letters, 483s, Recalls, Import Alerts, Audit observations
The Guidelines page guides you to important GMP and Regulatory guidance’s; Provides links to published final and draft guidelines of regulatory authorities like USFDA, EMA, WHO, MHRA, TGA and other NRAs. The page provides links to guidance’s and scientific discussion papers by expert bodies and technology platforms like ISPE, PICS, APIC, USP.
Guidance’s, literature and scientific discussion papers are collated subject wise to help navigate quickly to relevant document. Scroll over the titles to know which guidelines and topics are listed under the title. Click on + to open the list.
Qvents compilation of regulatory guidance’s on Computer System Validation, Electronic Data Review from USFDA, EMA, PIC-S. Access all guidelines on the topic – from USFDA 21 CFR Part 11 requirements, Questions and Answers, Document heavy CSV approach, Risk based frame work of CSA. Revised EC Annex 11 with focus on cybersecurity controls and EC Annex 22 focus on AI systems
Access all guidelines on the topic – from USFDA 21 CFR Part 11 requirements, Questions and Answers, Document heavy CSV approach, Risk based frame work of CSA. Revised EC Annex 11 with focus on cybersecurity controls and EC Annex 22 focus on AI systems
 Qvents compilation of regulatory guidelines on Data Integrity : FDA guidance for design, operation, and monitoring of systems and controls based on risk to patient, process, and product, Role of management, ALCOA++, meta data and audit trail, MHRA focus on organisational culture and data criticality based systems, WHO explanation of data governance and arrangements to provide assurance of data quality, Hybrid systems, Data Management and Pharma Quality Systems, PIC-S guidance, EMA and FDA requirements comparison, Data Integrity checklists
Qvents compilation of regulatory guidelines on Data Integrity : FDA guidance for design, operation, and monitoring of systems and controls based on risk to patient, process, and product, Role of management, ALCOA++, meta data and audit trail, MHRA focus on organisational culture and data criticality based systems, WHO explanation of data governance and arrangements to provide assurance of data quality, Hybrid systems, Data Management and Pharma Quality Systems, PIC-S guidance, EMA and FDA requirements comparison, Data Integrity checklists

Qvents compiled list of regulatory guidelines on GMP inspections from different agencies – USFDA, EMA, EDQM, WHO, PIC-S. Quick reference list:
What are different types of Inspections, GMP Inspection Process, Fees, Links to Inspection guides and checklists, Inspection outcomes, Regulatory actions, Consequences of denying, delaying, refusing GMP inspections, Post inspection remediations, Post Warning letter meetings, Useful presentations on GMP Inspections
EU GMP Annexure 16 Guidance’s, Qualified Person (QP), Batch Release, QP Declaration, Templates
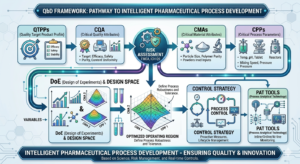
USFDA, EMA, ICH Guidelines on Pharmaceutical Development for Medicinal Products, Dosage Forms, Drug Substances (APIs), Quality by Design (QbD)
Regulatory Guidelines on Reserve Samples, Retention Samples, Reference Samples, Retention of Bioavailability and Bioequivalence (BA / BE) Samples, USFDA, EUGMP
Guidance’s and References for setting Specifications, Test methods, Acceptance criteria for Drug products (Formulations, Dosage Forms) and Drug substances (APIs) – Assay, Content, Impurity controls, Microbial controls, Elemental Impurities, Mutagenic/ Carcinogenic impurities, Nitrosamines, NDSRIs, Residual Solvents, General Characteristics; Pharmacopeia requirements: USP, EP; Regulatory guidelines: ICH, USFDA, EMA,
Hold time studies:

USP, EP, USFDA, EDQM requirements for Pharmaceutical Reference Standards, Secondary Standards, Qualification and traceability, WHO guidelines for Chemical Reference Substances, Characterisation and Qualification of Reference standards and Reference materials
Good Manufacturing Practices (GMP) guidance’s and regulatory requirements for Sterile manufacturing, Aseptic processing, Sterilisation Process control and Process validation, Aseptic process simulation / Media fill, Container Closure Integrity, Sterility assurance, Product sterility testing, Medicinal products, Active substances (APIs), Excipients, Primary containers – USFDA, EUGMP, EMA, WHO, PICS, PDA, USP
 USFDA : Sterile Drug Products Produced by Aseptic Processing — Current Good Manufacturing Practice – Guidance
USFDA : Sterile Drug Products Produced by Aseptic Processing — Current Good Manufacturing Practice – Guidance
Regulatory guidelines on Pharmaceuticals Storage and distribution, Temperature controlled storage, Temperature mapping, Temperature cycling studies, Transporter qualification and validation, Good storage and shipping practices – WHO, PDA, TGA, Health Canada, ANVISA, USP guidance’s and requirements
Regulatory guidance’s on Pharmaceutical Technology Transfer: WHO Guidelines, ISPE Good Practices PDA Technical Report, IPA (India) guidance